Want to speak with one of our Product experts?
available Mon-Fri 9AM-3PM Arizona Time
Want to speak with one of our Product experts?
available Mon-Fri 9AM-3PM Arizona Time
Since 1982 PhosChol PPC is the #1 Selling Healthcare Practitioner Brand
pharmaceutical grade | therapeutically dosed | professionally formulated
Are you missing the one essential ingredient that is absolutely critical to achieving your best possible health and well-being? If you are not already taking PPC then the answer is YES!
According to Dr. Sherry Rogers MD, a very well-respected authority on alternative medicine and nutrition, PPC is perhaps the most important ingredient you should take to reach your next level of health. In her book Detox or Die, she rightly points out that because PPC is such a meaningful part of the cell membrane structure and functioning, it is the one reason why some people will never get better...and that's because "no one ever recommended it".
Life Happens on the Cell Membrane
Life as we know it happens on our cell membranes. We are each nothing more than ~50 trillion cells held together by some sort of cosmic glue. And without a cell membrane to form our cells, we wouldn't be alive at all. The cell membrane is that essential and foundational to life; and so, it makes sense that if you want to be your healthiest self, you must start with the health of your cell membranes.
You cannot build a house on top of a compromised and unstable foundation, and you cannot achieve better health on top of compromised and unstable cell membranes. It's really that simple!
Stuck on a Health and Healing Plateau?
If you are stuck on a health and healing plateau, or perhaps just struggling a little with low energy and brain fog, the answer may be in the poor health and functioning of your cell membranes.
Luckily for us there is PPC, a tried-and-true molecule that helps get our cell membranes working the way they should. This molecule is so essential that some scientists have even referred to it as the "miracle molecule".
PPC may be just what you need to jump start your good health and anti-aging journey.
Stuck on a Health and Healing Plateau?
If you are stuck on a health and healing plateau, or perhaps just struggling a little with low energy and brain fog, the answer may be in the poor health and functioning of your cell membranes.
Luckily for us there is PPC, a tried-and-true molecule that helps get our cell membranes working the way they should. This molecule is so essential that some scientists have even referred to it as the "miracle molecule".
PPC may be just what you need to jump start your good health and anti-aging journey.
PhosChol is the Only Pharmaceutical Grade PPC
PPC is an essential building block for life and better health! PPC goes to work throughout the whole body to restructure, repair and rejuvenate cell membranes damaged by inflammation and oxidative stress. Every cell and organelle membrane benefits from PPC and with-it cell structure, function and metabolism is restored. PPC is holistic and therapeutic, and its applications are clinically significant.
PPC is particularly active and beneficial:
-
As a Holistic Cell Membrane Therapy*
- Supports Gut Health and Repair*
- Enhances Acetylcholine Synthesis & Neurological Processes *
-
Increases Glutathione Production in the Liver Naturally by Restoring Depleted SAMe (S-Adenosyl-Methionine) Levels*
-
Supports Liver Cell Membrane Restructuring, Repair, Regeneration and Proliferation*
-
An Essential Building Block for Life and a Critical Component to Foundational Whole Body Health*
PURE PCC
A Superior Cell Membrane Therapeutic
Improve your health with PhosChol PPC. A superior cell membrane therapeutic, PPC acts primarily on cellular repair, regeneration, proliferation and metabolism, with special emphasis on vulnerable organs such as the liver and brain. Based on over 70 years of extensive clinical research, PPC plays a critical role in the healthy function of every cell and organelle (i.e. Mitochondria) in your body*
PPC makes everything you're taking work better

Since 1982 PhosChol PPC is the #1 Selling Healthcare Practitioner Brand
pharmaceutical grade therapeutically dosed professionally formulated
Are you missing the one essential ingredient that is absolutely critical to achieving your best possible health and well-being? If you are not already taking PPC then the answer is YES!
According to Dr. Sherry Rogers MD, a very well-respected authority on alternative medicine and nutrition, PPC is perhaps the most important ingredient you should take to reach your next level of health. In her book Detox or Die, she rightly points out that because PPC is such a meaningful part of the cell membrane structure and functioning, it is the one reason why some people will never get better...and that's because "no one ever recommended it".
Life Happens on the Cell Membrane
PhosChol is the Only Pharmaceutical Grade PPC
PPC is an essential building block for life and better health! PPC goes to work throughout the whole body to restructure, repair and rejuvenate cell membranes damaged by inflammation and oxidative stress. Every cell and organelle membrane benefits from PPC and with-it cell structure, function and metabolism is restored. PPC is holistic and therapeutic, and its applications are clinically significant.
PPC is particularly active and beneficial:
-
As a Holistic Cell Membrane Therapy*
- Supports Gut Health and Repair*
- Enhances Acetylcholine Synthesis & Neurological Processes *
-
Increases Glutathione Production in the Liver Naturally by Restoring Depleted SAMe (S-Adenosyl-Methionine) Levels*
-
Supports Liver Cell Membrane Restructuring, Repair, Regeneration and Proliferation*
-
An Essential Building Block for Life and a Critical Component to Foundational Whole Body Health*
PURE PPC
A Superior Cell Membrane Therapeutic
Improve your health with PhosChol PPC. A superior cell membrane therapeutic, PPC acts primarily on cellular repair, regeneration, proliferation and metabolism, with special emphasis on vulnerable organs such as the liver and brain. Based on over 70 years of extensive clinical research, PPC plays a critical role in the healthy function of every cell and organelle (i.e. Mitochondria) in your body*
PPC makes everything you're taking work better

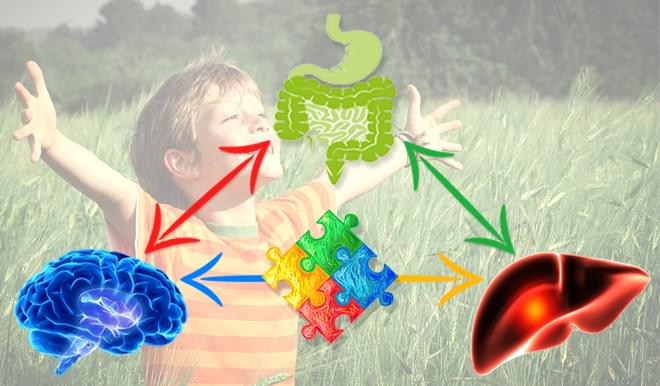
How You Will Benefit from Taking PPC
The benefits of PPC are broad and deep. Billions of doses have been administered globally since 1953 and since 1982, over 200 million doses have been administered by Doctors in the United States Alone. The list of positive physiological changes in the body resulting from PPC administration is exhaustive. The benefits described here in the "Macro Environment" only scratch the surface of the full reach and benefit of PPC administration.
PPC In Cell Membranes
Life happens on the cell membrane and purified PPC provides the building blocks essential to cellular membrane formation, restructure, repair, regeneration, proliferation and function. Like bricks in a wall, cellular membranes act as barriers to protect each cell and organelle in the human body, maintaining cellular structure, function and co-operation. Membranes are an essential building block for life as cells, tissues, organs and ultimately an organism cannot be formed without them. Without PC and cellular membranes, life as we know it would not be possible.

PolyenylPhosphatidylcholine (PPC) clearly plays a vital role in human life. Its role is even more important when unhealthy lifestyle choices are made. For example, excess consumption of processed carbohydrates, sugar and HFCS, alcohol, and drugs and prescription medications, among others, can lead to cell membrane damage. When cell membranes become oxidized and damaged, cell metabolism and function decreases, and good health becomes compromised. When membrane damages are present, PPC is in high demand to help form, proliferate, restructure and repair cell membranes and to enhance cell membrane-dependent metabolism.

PPC In The Liver
PPC preferentially supports our overworked and underappreciated liver. Over 200 clinical studies demonstrate the importance of PPC in supporting healthy liver function. In the liver PPC restructures damaged cell membranes, normalizes cellular permeability, restores SAMe, improves transmethylation, transsulfuration, increases glutathione production and supports healthy liver detox and function. PPC acts antioxidatively in the liver by reducing toxic free radicals and by replenishing the body's own glutathione pool.

PPC In Mitochondria
Mitochondria are well known as the energy-producing powerhouses of our cells. However, they cannot generate energy through the electron transport chain without healthy and intact membranes. The integrity of mitochondrial membranes is critical to cell function and energy metabolism. PPC positively effects the structure and function of mitochondria by improving membrane ultrastructure, respiration, energization and anti-apoptotic activity.

PPC In Blood Vessels
PPC positively influences various conditions related to the development and progression of plaque formation, such as lipoprotein alterations and free radical-induced lipid peroxidation. Other results include healthy cholesterol, triglyceride, and LDL to HDL cholesterol ratios. PPC also supports healthy blood flow properties and improvements in microcirculation. In order to be effective an active solution should act simultaneously on the blood and on the vascular walls. PPC meets this requirement.

PPC in Immune System
The discovery that the administration of PPC leads to a series of complex mechanisms triggering Natural Killer Cell Activity and the enhancement of phagocytosis and "killing” by PMNs is significant. This discovery has led researchers to conclude that “clearly the dramatic effect of PPC on the clinical course and outcome of both bacterial and viral infections empirically demonstrates the use and value of PPC and related molecules as effective therapeutic and prophylactic agents for bacterial, yeast protist and
viral infections."
PPC In The Brain
PPC provides a biologically preferred source of choline that is safe and readily available for use in the body's manufacture of acetylcholine, an essential neurotransmitter. PPC increases cell membrane fluidity and integrity, restructures and repairs damaged neuronal cell membranes, reactivates membrane bound enzymes & receptors. PPC acts antioxidatively and by doing so helps to reduce neuro inflammation. PPC provides essential support for neurological development and brain function.

PPC In The Gut
PPC supplies the gut with readymade surfactant material to support the gastrointestinal mucosal barrier. PPC is an essential component of intestinal mucus and is greater than 90% of all available phospholipids. Orally supplemented PPC has been shown to support the formation of surfactant like particles which provide a protective hydrophobic surface on top the hydrated mucus gel that prevents the invasion of bacteria from intestinal lumen and the corresponding inflammation. *

PPC In Glutathione
PPC increases glutathione production naturally and replenishes the body’s own Glutahtione (GSH) pool. The probable reason is that PPC corrects the hepatic oxidative stress by restoring depleted S-adenosylmethionine (SAMe), and adequate levels of SAMe are necessary to produce enough GSH by transsulfuration. GSH is a major intracellular antioxidant and increased levels of GSH are necessary to detoxify the liver, fight against toxic radicals and reduce oxidative stress.

PPC in Skin & Aging
Skin and other membranes change composition during the aging process, as more cholesterol and less phosphatidylcholine with polyunsaturated fatty acids are incorporated into the membranes. They frequently become more rigid, partly recognized as wrinkles. PPC restructures cell membranes, increases membrane flexibility, repairs cell and organelle membranes and improves cell metabolism and function. In short, PPC keeps cells younger, healthier, and more active.

How You Will Benefit from Taking PPC
The benefits of PPC are broad and deep. Billions of doses have been administered globally since 1953 and since 1982, over 200 million doses have been administered by Doctors in the United States Alone. The list of positive physiological changes in the body resulting from PPC administration is exhaustive. The benefits described here in the "Macro Environment" only scratch the surface of the full reach and benefit of PPC administration.
PPC In Cell Membranes
Life happens on the cell membrane and purified PPC provides the building blocks essential to cellular membrane formation, restructure, repair, regeneration, proliferation and function. Like bricks in a wall, cellular membranes act as barriers to protect each cell and organelle in the human body, maintaining cellular structure, function and co-operation. Membranes are an essential building block for life as cells, tissues, organs and ultimately an organism cannot be formed without them. Without PC and cellular membranes, life as we know it would not be possible.


PPC In The Liver
PPC preferentially supports our overworked and underappreciated liver. Over 200 clinical studies demonstrate the importance of PPC in supporting healthy liver function. In the liver PPC restructures damaged cell membranes, normalizes cellular permeability, restores SAMe, improves transmethylation, transsulfuration, increases glutathione production and supports healthy liver detox and function. PPC acts antioxidatively in the liver by reducing toxic free radicals and by replenishing the body's own glutathione pool.

PPC In Mitochondria
Mitochondria are well known as the energy-producing powerhouses of our cells. However, they cannot generate energy through the electron transport chain without healthy and intact membranes. The integrity of mitochondrial membranes is critical to cell function and energy metabolism. PPC positively effects the structure and function of mitochondria by improving membrane ultrastructure, respiration, energization and anti-apoptotic activity.

PPC In Blood Vessels
PPC positively influences various conditions related to the development and progression of plaque formation, such as lipoprotein alterations and free radical-induced lipid peroxidation. Other results include healthy cholesterol, triglyceride, and LDL to HDL cholesterol ratios. PPC also supports healthy blood flow properties and improvements in microcirculation. In order to be effective an active solution should act simultaneously on the blood and on the vascular walls. PPC meets this requirement.

PPC in The Immune System
The discovery that the administration of PPC leads to a series of complex mechanisms triggering Natural Killer Cell Activity and the enhancement of phagocytosis and "killing” by PMNs is significant. This discovery has led researchers to conclude that “clearly the dramatic effect of PPC on the clinical course and outcome of both bacterial and viral infections empirically demonstrates the use and value of PPC and related molecules as effective therapeutic and prophylactic agents for bacterial, yeast protist and
viral infections."
PPC In The Brain
PPC provides a biologically preferred source of choline that is safe and readily available for use in the body's manufacture of acetylcholine, an essential neurotransmitter. PPC increases cell membrane fluidity and integrity, restructures and repairs damaged neuronal cell membranes, reactivates membrane bound enzymes & receptors. PPC acts antioxidatively and by doing so helps to reduce neuro inflammation. PPC provides essential support for neurological development and brain function.

PPC In The Gut
PPC supplies the gut with readymade surfactant material to support the gastrointestinal mucosal barrier. PPC is an essential component of intestinal mucus and is greater than 90% of all available phospholipids. Orally supplemented PPC has been shown to support the formation of surfactant like particles which provide a protective hydrophobic surface on top the hydrated mucus gel that prevents the invasion of bacteria from intestinal lumen and the corresponding inflammation. *

PPC In Glutathione
PPC increases glutathione production naturally and replenishes the body’s own Glutahtione (GSH) pool. The probable reason is that PPC corrects the hepatic oxidative stress by restoring depleted S-adenosylmethionine (SAMe), and adequate levels of SAMe are necessary to produce enough GSH by transsulfuration. GSH is a major intracellular antioxidant and increased levels of GSH are necessary to detoxify the liver, fight against toxic radicals and reduce oxidative stress.
PPC in Skin & Aging
Skin and other membranes change composition during the aging process, as more cholesterol and less phosphatidylcholine with polyunsaturated fatty acids are incorporated into the membranes. They frequently become more rigid, partly recognized as wrinkles. PPC restructures cell membranes, increases membrane flexibility, repairs cell and organelle membranes and improves cell metabolism and function. In short, PPC keeps cells younger, healthier, and more active.

What People Are Saying About PhosChol PPC
Thousands of Testimonials Since 1982
These are just a few
What People Are Saying About PhosChol PPC
Thousands of Testimonials Since 1982
These are just a few

Ben
Great Product and Service Have been taking this daily for at least 6 mos. Cholesterol profile was improved compared to last year. HDL:LDL ratio better. Product was well packaged and shipped fast
Kristen
Great Product. I have been using PPC for some time and have felt a difference since taking it. I only take 1 capsule a day to help minimize the formation of gallstones. The pain in my upper right abdomen has decreased significantly since I started taking it. It seems to be the highest quality form of phosphatidylcholine on the market.
Lynn
Great product and vegetarain. I think nutrasal ppc is the best product and as a vegan it is great to have it in vegan friendly capsule. I take it for anti-aging/brain and liver and would recommend this product.
Wayne
My son has autism. He and I have been taking phoschol for over 20 years. He absolutely loves it and must have it. He feels and functions better. When we run out, it's not good. I swear by this product and highly recommend it.
CUSTOM JAVASCRIPT / HTML
Better Health & Better Results Start with PPC
- Over 40 years of use in the United States
- Unique mode of action and proven results
- Broad and deep clinical documentation
- Proven safe and effective
- 100% Satisfaction and Happiness Guaranteed
Experience the Difference PhosChol PPC can have on your health and in the way you feel. More energy and better focus is what you'll feel, and better health is what you'll get. You're body will thank you for taking PhosChol.
There's a reason PhosChol PPC is the leading healthcare practitioner brand for over 40 years, and that reason is quite simple...it's absolutely essential and it works!
PhosChol is the Gold Standard
# 1 Selling Healthcare Practitioner Brand
Over 70 years of clinical studies and pharmacological investigations demonstrate PPC is an essential and superior cell membrane therapeutic.
PhosChol PPC is the holistic and physiological way to better health and wellness.
In numerous clinical studies, purified PPC has been shown to support:*
- Cardiovascular health
- Liver health and function
- Brain Function and memory
- Cell membrane restructure, repair and rejuvenation, and improve cell metabolism and function
-
Gastric mucosa protection
- Glutathione production increases and enhanced cell and liver detoxification
-
Immune System and NK cell activity
-
Sports Endurance and Recovery
-
Among others
Recommended by Doctors more than any other Brand
Phoschol is presented in multiple capsule and liquid forms.
- Phoschol Gelatin Capsules (30ct, 100ct)
- PhosChol Vegetable Capsules (60ct, 120ct)
- PhosChol Liquid Concentrate (8oz, 16oz)
- PhosChol PPC Kids Liposomal Orange Cream (8oz)

Better Health & Better Results Start with PPC
Experience the Difference PhosChol PPC can have on your health and in the way you feel. More energy and better focus is what you'll feel, and better health is what you'll get. You're body will thank you for taking PhosChol.
There's a reason PhosChol PPC is the leading healthcare practitioner brand for over 40 years, and that reason is quite simple...it's absolutely essential and it works!
PhosChol is the Gold Standard
# 1 Selling Healthcare Practitioner Brand
Over 70 years of clinical studies and pharmacological investigations demonstrate PPC is an essential and superior cell membrane therapeutic.
PhosChol PPC is the holistic and physiological way to better health and wellness.
In numerous clinical studies, purified PPC has been shown to support:*
- Cardiovascular health
- Liver health and function
- Brain Function and memory
- Cell membrane restructure, repair and rejuvenation, and improve cell metabolism and function
-
Gastric mucosa protection
- Glutathione production increases and enhanced cell and liver detoxification
-
Immune System and NK cell activity
-
Sports Endurance and Recovery
-
Among others
Recommended by Doctors more than any other Brand
Phoschol is presented in multiple capsule and liquid forms.
- Phoschol Gelatin Capsules (30ct, 100ct)
- PhosChol Vegetable Capsules (60ct, 120ct)
- PhosChol Liquid Concentrate (8oz, 16oz)
- PhosChol PPC Kids Liposomal Orange Cream (8oz)

Why PhosChol PPC?
PhosChol - The #1 Selling Practitioner Brand since 1982 and the "Best Source of Purifed PPC"
Well over 200 million doses of PhosChol have been administered by doctors in the United States alone. PPC is Proven to be Safe and Very Effective.
PhosChol is the 100 percent purified and pharmaceutical grade PPC that delivers the highest concentrated source of 1,2 DLPC...the special and primary active molecule, which accounts for its superior health benefits and outstanding results. If you want to look and feel your best PPC will help you get there.*
Why PhosChol PPC?
PhosChol - The #1 Selling Practitioner Brand since 1982 and the "Best Source of Purifed PPC"
Well over 200 million doses of PhosChol have been administered by doctors in the United States alone. PPC is Proven to be Safe and Very Effective.
PhosChol is the 100 percent purified and pharmaceutical grade PPC that delivers the highest concentrated source of 1,2 DLPC...the special and primary active molecule, which accounts for its superior health benefits and outstanding results. If you want to look and feel your best PPC will help you get there.*
Purchase Using Your Health Saving Account (HSA)

Great News!
HSA & FSA Cards Now Accepted!
You can now purchase our professionally formulated products using your HSA or FSA Health Spending account. No Special Requirements are Necessary. You can simply use your card the way you would your Visa, Mastercard or American Express.
The best part about using your HSA or FSA Card is you get to utilize your pre-tax savings to purchase nutritional health products. This means you can save up to 37%. Savings are based on your tax bracket at filing.
We do not require any paperwork or a doctor’s referral in order to process your purchase. You can just add your items to your shopping cart and check out using your HSA Card. It’s that Simple.
You may want to check with your HSA/FSA account provider to see if they require any documentation or record keeping for your supplement purchases. And you may want to have your doctor’s referral on file in case of an IRS audit. We have additional information below to assist you in making your HSA or FSA purchases.
HSA/FSA Guide Reimbursement Form
The Nutrasal information guide and reimbursement form is provided for your convenience. It is not tax or legal advice. For more information about using your HSA Card for dietary supplements please visit www.irs.gov or your HSA Provider.

Purchase Using Your Health Saving Account (HSA)

Great News!
HSA & FSA Cards Now Accepted!
You can now purchase our professionally formulated products using your HSA or FSA Health Spending account. No Special Requirements are Necessary. You can simply use your card the way you would your Visa, Mastercard or American Express.
The best part about using your HSA or FSA Card is you get to utilize your pre-tax savings to purchase nutritional health products. This means you can save up to 37%. Savings are based on your tax bracket at filing.
We do not require any paperwork or a doctor’s referral in order to process your purchase. You can just add your items to your shopping cart and check out using your HSA Card. It’s that Simple.
You may want to check with your HSA/FSA account provider to see if they require any documentation or record keeping for your supplement purchases. And you may want to have your doctor’s referral on file in case of an IRS audit. We have additional information below to assist you in making your HSA or FSA purchases.
HSA/FSA Guide Reimbursement Form
The Nutrasal information guide and reimbursement form is provided for your convenience. It is not tax or legal advice. For more information about using your HSA Card for dietary supplements please visit www.irs.gov or your HSA Provider.

Need Additional Information?
Please feel free to call us for a product consult. We're happy to assist you in any way we can.
Purified PPC (Phosphatidylcholine - Pharmaceutical Grade) has been studied extensively since the 1950's and PhosChol PPC is the only dietary supplement to contain the all-natural oilseed medicinal found as the primary active ingredient in 2 registered pharmaceutical products. (Source: National Resource Council - Ottawa, Canada)
You can find more information regarding this fundamentally essential phospholipid by visiting our website www.nutrasal.com

Need Additional Information?
Please feel free to call us for a product consult. We're happy to assist you in any way we can.
Purified PPC (Phosphatidylcholine - Pharmaceutical Grade) has been studied extensively since the 1950's and PhosChol PPC is the only dietary supplement to contain the all-natural oilseed medicinal found as the primary active ingredient in 2 registered pharmaceutical products. (Source: National Resource Council - Ottawa, Canada)
You can find more information regarding this fundamentally essential phospholipid by visiting our website www.nutrasal.com


Nutrasal, Inc | Copyright ©2022 | All Rights Reserved